 
All that you have to do is substitute these values in the above formula. The number of electrons in non-bonded state are 2, while the ones in the covalent bond are 8. The number of valence electrons of carbon are 4. Here, carbon is bonded with four hydrogen atoms. 
Let me now illustrate the calculation for a carbon atom in methane molecule (CH 4). The right Lewis structure will be the one for which the sum of formal charges has a minimum value. To be able to decide which is the most stable Lewis structure for any compound, one must calculate the formal charge of every atom involved in the compound and sum up all the values. In this formula, V stands for the number of valence electrons of that atom ( these are the electrons that revolve in the outermost orbit of the atom), N stands for the number of non-bonded electrons, and B stands for the number of electrons that are a part of the covalent bonds made by the atom. So now you have only the number of atom that are. It is the difference between the valence electron number of the atoms and the combined sum of bonded and non-bonded electrons in an atom. You take all the possible atom that can bond then subtract the number of lone pairs. 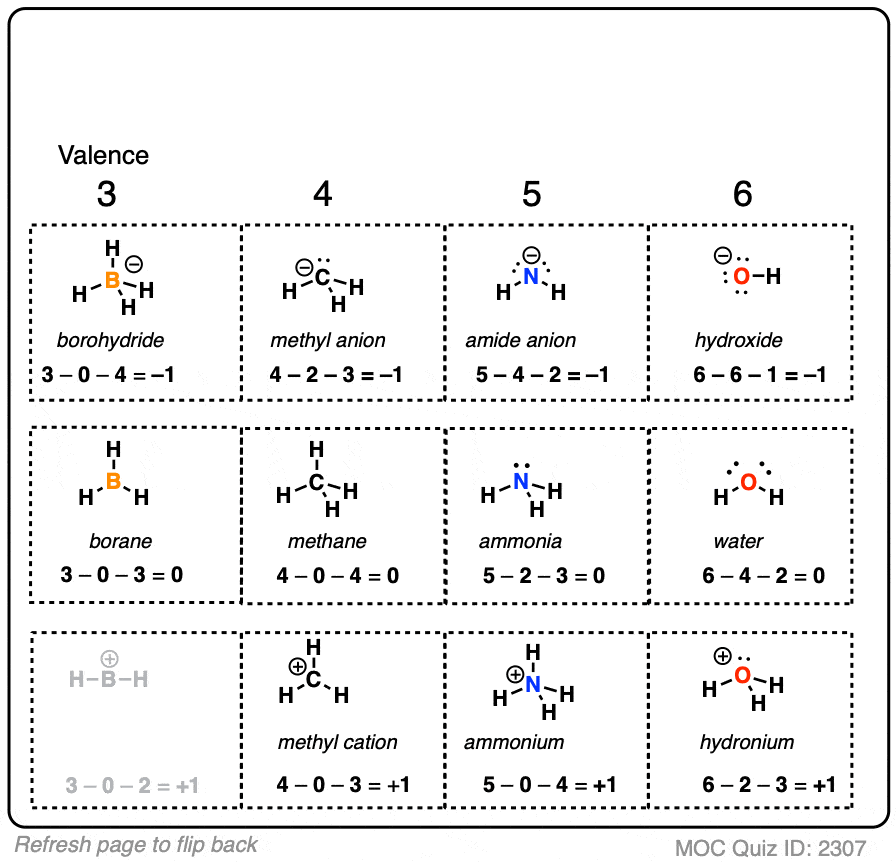
Formal charge calculation enables you to determine this. This necessitates that a chemist knows the low energy electronic configuration for every one of the bonding atoms. formal charge kernel charge - assigned electrons calculate by assigning electrons to each atom lone pairs belong completely to their parent atom bonding. When determining the exact molecular structure, one needs to know how the molecules have bonded together. Nature prefers low energy states which lead to stable molecular structures. DefinitionĪtoms combine together to form molecules by sharing electrons ( forming a covalent bond) or by exchanging electrons ( through an ionic bond). Now each Cl atom has seven electrons and the Br atom has seven. Assign one of the electrons in each BrCl bond to the Br atom and one to the Cl atom in that bond: Step 2. 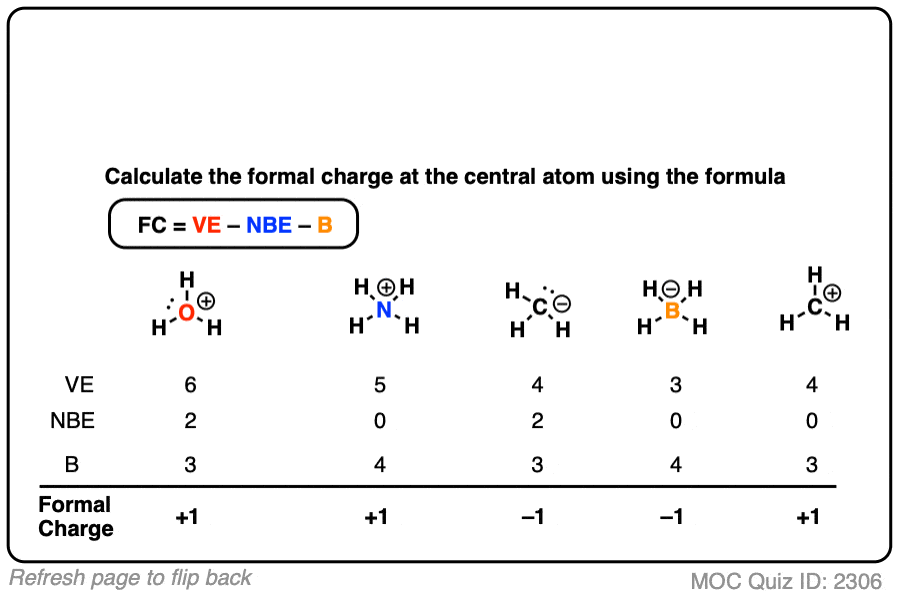
Another pictorial technique for depicting the electronic distribution and bonding of a molecule is a Lewis structure. Calculating Formal Charge from Lewis Structures Assign formal charges to each atom in the interhalogen molecule BrCl 3. Molecular nomenclatures are developed to accurately describe the structural details and expanse of any molecule. A large part of chemistry study is about learning to identify and visualize molecules. You need to be adept at experimenting and theorizing to be a chemist. Generally, the lowest energy structure is the one with the smallest formal charges on the atoms and the most distributed charge.What I like about chemistry is the amazing amount of detective work that is involved in determining the actual structure of a molecule.Knowledge of the lowest energy structure helps in predicting the major product of a reaction and also describes a lot of phenomena.Formal charges help in the selection of the lowest energy structure from a number of possible Lewis structures for a given species.The formal charge being a theoretical charge doesn’t indicate any real charge rather separation in the molecule.A negative formal charge indicates that an atom is more likely to be the source of electrons while a positive one, on the other hand, it is more likely to take electrons, and most likely be the reaction’s site. The formal charge of a molecule can indicate how it will behave during a process. This condition could point to resonance structures, especially if the structures have the same atom arrangement but different types of arrangements of bonds. While formal charge can indicate a molecule’s preferred structure, the problem becomes more complicated when numerous equally preferred structures exist. The one with the least/lowest formal charges is the ideal structure. An atom in a molecule should have a formal charge of zero to have the lowest energy and hence the most stable state. Knowing the formal charge on a particular atom in a structure is an important part of keeping track of the electrons and is important for establishing and predicting the reactivity. Prior to discussing how to calculate formal charge(FC), we must understand that it is n essential, basic concept to master in order to better understand molecular structures and reactionsīasically, we can define formal charge as the hypothetical measure of charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
